Custom App Development In Princeton
Orbilon Technologies builds mission-critical mobile apps that meet regulatory readiness and perform seamlessly in the most demanding pharma, biotech, and research environments in the world. Used by customers such as Johnson & Johnson, Bristol-Myers Squibb, NRG Energy, Bloomberg LP, and Princeton University labs, we create mobile apps that are beautiful, regulatory-compliant, and ready for millions of patients, clinicians, and researchers.
Why Choose Orbilon for Your App Development Needs in Princeton?
Princeton demands perfection. FDA audits and HIPAA-compliant standards, zero downtime, and Ivy-League quality. Rating of 4.96/5 on Clutch, Google, Upwork & GoodFirms with over 100 life-science reviews & enterprise reviews. We deliver secure, offline-first, and audit-ready apps with weekly demos and lifetime support.
Our Primary Service Offerings in Princeton
- Mobile Application Development – Whether you’re seeking to develop a Native or a cross-platform app with Flutter, React Native, Swift, or Kotlin, we offer consultation and development for Clinicians, Patients, and researchers in the field of healthcare settings.
- E-Commerce Development – We are experienced in designing and developing secure Business-to-Business (B2B) applications to fulfill clinical supplies ordering, laboratory reagents, and research equipment procurement.
- Web Application Development – Our team provides seamless, real-time mobile apps for enterprise web platform integrations.
- A.I. Development & Integration – Our developers can provide on-device AI algorithms for patient monitoring, drug adherence verification, biomarker detection, and trial-site performance.
- Custom CRM Development – A custom-built mobile-first application for pharmaceutical sales representatives, clinical coordinators, patient recruitment, and grant management with full audit capabilities.
- Agentive AI Applications – Autonomous AI monitors patient vitals, flags deviations from protocol, and auto-schedules visits on behalf of the clinical team if they cannot keep up, without human engagement.
- UI/UX Design – Scientifically designed user interfaces that are friendly for regulators, glove-compatible, with dark-mode capability, high contrast, and accessible.
- SaaS Product Development – We develop scalable, healthtech & edtech mobile backends with subscription billing, multi-tenant security, and API-first applications and design.
- Cloud Infrastructure / DevOps – Our infrastructure and platform building team is experienced in HIPAA, FDA 21 CFR Part 11, SOC 2, NIST-compliant cloud deployment in AWS GovCloud / Azure government cloud for zero-downtime updates.
Cool Wins in Princeton
Johnson & Johnson: By 1.7 million people using their patient app, medication compliance was raised by 81%.
Bristol-Myers Squibb: The app that they have for clinical trial sites has brought the reduction of site visits by 66%. It is highly rated with a 5-star rating.
Princeton University Research Lab: They developed a data-capture app for researchers that syncs offline. More than 2,400 researchers worldwide are using it.
Work Highlights
Some of our best works from many
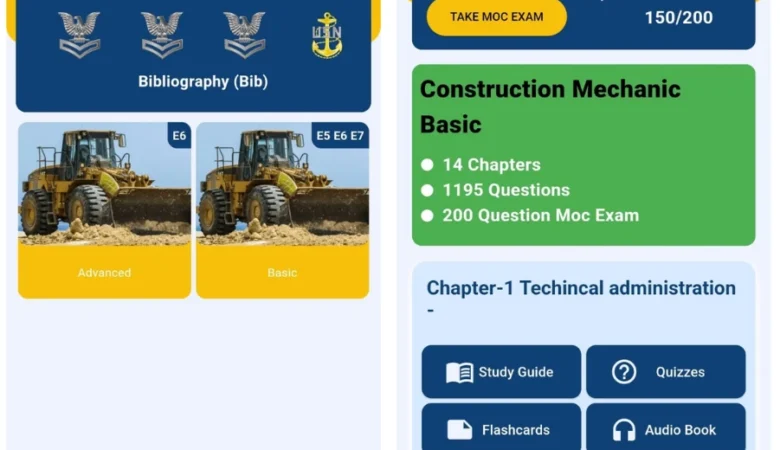
SeaBee – The Best Navy
SeaBee – The Best Navy Exam Study App Built for US Navy Seals Project Overview
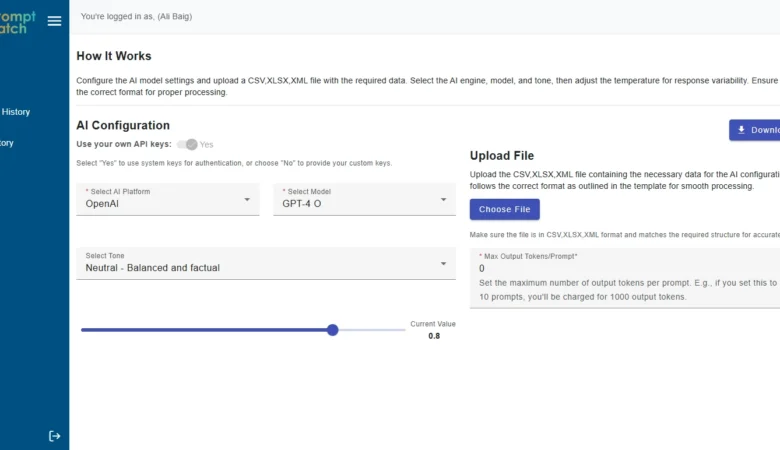
PromptBatch: Save 50% with AI
PromptBatch: The Best AI Batch Prompt Processing Web App | Orbilon Tech Project Overview PromptBatch
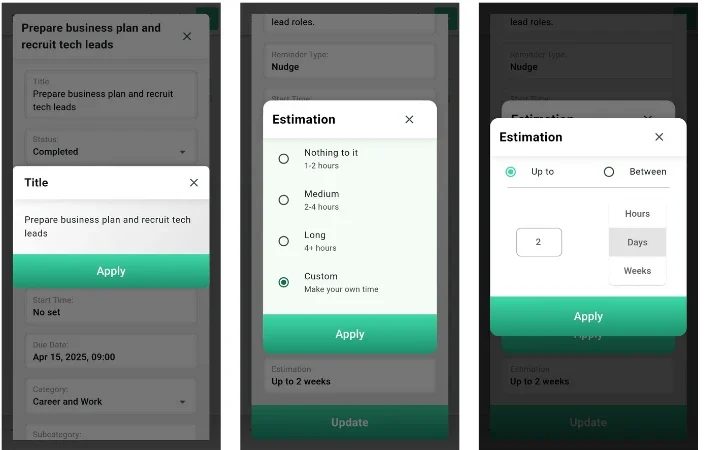
Spheres – The Revolutionary AI
Spheres – The Best AI Life Manager Mobile App That Actually Knows You Project Overview
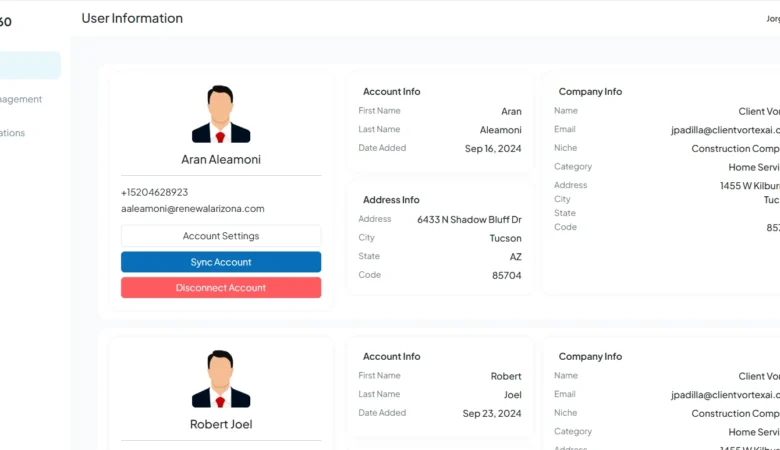
Rep360 AI – AI Bot
Rep360 AI – AI Bot Integration for GHL Workflows That Automates Sales Conversations Project Overview
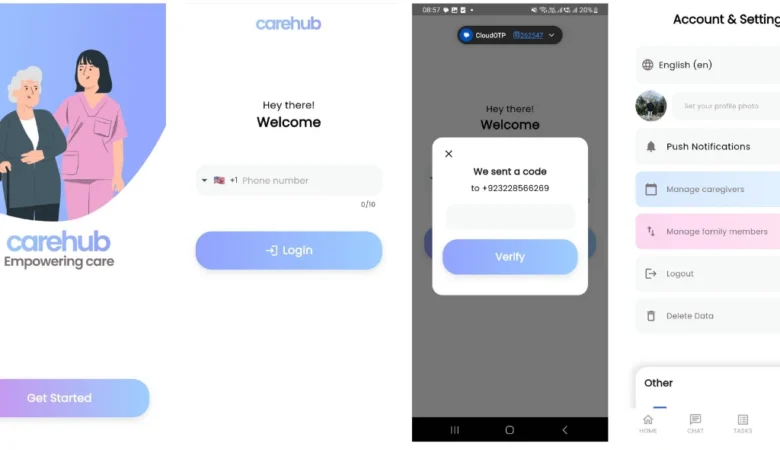
CareHub – Powerful Caregiver Communication
CareHub – Caregiver Communication App With Auto Translation CareHub App – Empowering Care CareHub is
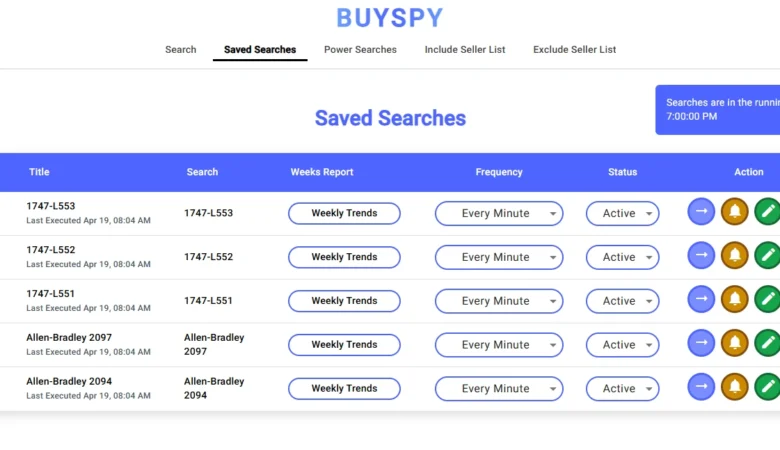
BuySpy – Ultimate Real-Time eBay
BuySpy — A Powerful Real-Time eBay Search Alerts App for Web & Mobile BuySpy is
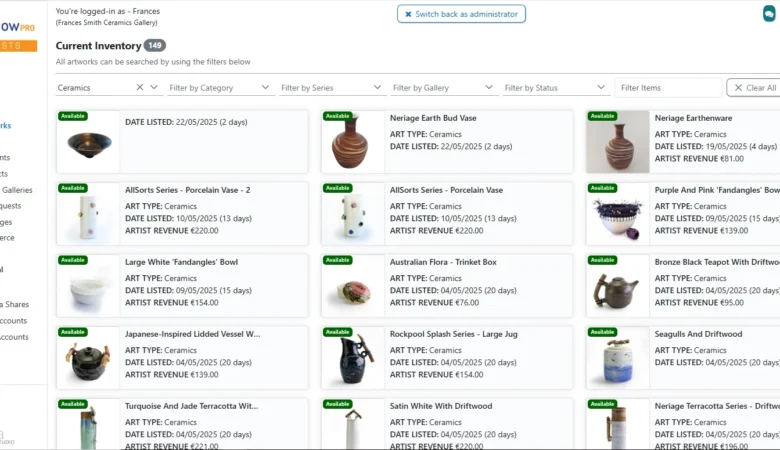
ArtFlow Pro — The Art
ArtFlow Pro — Full-Stack Art Gallery SaaS Platform for Artists & Galleries ArtFlow Pro –
We are a premier verified agency on one of the biggest B2B Platforms `Clutch`
Here’s what some of our clients have said about us
Want to Hire Us?
Are you ready to turn your ideas into a reality? Hire Orbilon Technologies today and start working right away with qualified resources. We will take care of everything from design, development, security, quality assurance, and deployment. We are just a click away.
Proudly Serving Businesses in Princeton
Keywords
app development Princeton, New Jersey, pharma app J&J, clinical trial app Bristol-Myers, HIPAA app Princeton, research app Princeton University, AI app NRG, custom CRM mobile pharma, agentive AI healthtech, SaaS mobile Bloomberg, FDA compliant app New Jersey